Computer Systems Validations Testing Services
Computer Systems Validations Testing Services Specification
- Expertise
- CSV Experts with domain knowledge
- Service Mode
- Online/Offline
- Documentation Provided
- Validation protocols, Test reports, SOPs
- Location
- Pan India
- Usage/Application
- Software Systems, IT Infrastructure, Regulated Industries
- Confidentiality
- Strict adherence to client confidentiality
- Testing Tools Used
- Customized and industry-standard tools
- Duration
- Project Based
- Testing Type
- Validation Testing
- Compliance Standards
- 21 CFR Part 11, GAMP 5, Annex 11
- Support
- End-to-end validation lifecycle support
- Industries Served
- Pharmaceutical, Healthcare, Biotechnology, Manufacturing
- Service Type
- Computer Systems Validations Testing Services
Computer Systems Validations Testing Services Trade Information
- Minimum Order Quantity
- 1 Number
- Payment Terms
- Cash in Advance (CID)
- Supply Ability
- 10 Per Month
- Delivery Time
- 5-7 Days
- Main Domestic Market
- All India
About Computer Systems Validations Testing Services
Experience exclusive Computer Systems Validations (CSV) Testing Services, engineered for compliance with 21 CFR Part 11, GAMP 5, and Annex 11. Our advance purchase packages ensure the lowest price for unprecedented quality, backed by meritorious protocols and robust reporting. Serving pharmaceutical, healthcare, biotechnology, and manufacturing sectors across India, our first-string team of CSV experts leverages both customized and industry-standard testing tools. We provide rigorous validation documentation and uphold strict client confidentiality, supporting every project with end-to-end services both online and offline.
Commercial Applications and Versatile Media Use
Our CSV Testing Services are indispensable for regulated industries, including pharmaceuticals, healthcare, biotechnology, and manufacturing. Designed for versatile application media, such as software systems and IT infrastructure, our services feature comprehensive documentation-validation protocols, SOPs, and test reports. Leveraging our expertise not only ensures regulatory compliance but also boosts operational efficiency. The service offers custom and standard tools, fostering quality assurance in software and systems essential for critical commercial applications.
Supply Capacity, Sample Policies, and Delivery Timelines
We maintain robust supply ability, capable of handling multiple concurrent projects efficiently, ensuring timely handover of validation deliverables. Our sample policy allows potential clients to review initial documentation prior to engagement, providing assurance of service standards before any outlay. Delivery timeframes are optimized for each project, with efficient goods transport and coordinated logistics, ensuring seamless execution and punctual delivery across various locations throughout India.
Commercial Applications and Versatile Media Use
Our CSV Testing Services are indispensable for regulated industries, including pharmaceuticals, healthcare, biotechnology, and manufacturing. Designed for versatile application media, such as software systems and IT infrastructure, our services feature comprehensive documentation-validation protocols, SOPs, and test reports. Leveraging our expertise not only ensures regulatory compliance but also boosts operational efficiency. The service offers custom and standard tools, fostering quality assurance in software and systems essential for critical commercial applications.
Supply Capacity, Sample Policies, and Delivery Timelines
We maintain robust supply ability, capable of handling multiple concurrent projects efficiently, ensuring timely handover of validation deliverables. Our sample policy allows potential clients to review initial documentation prior to engagement, providing assurance of service standards before any outlay. Delivery timeframes are optimized for each project, with efficient goods transport and coordinated logistics, ensuring seamless execution and punctual delivery across various locations throughout India.
FAQ's of Computer Systems Validations Testing Services:
Q: How do Computer Systems Validations Testing Services ensure regulatory compliance?
A: Our services strictly adhere to industry standards such as 21 CFR Part 11, GAMP 5, and Annex 11, providing comprehensive documentation and rigorous validation testing to meet regulatory requirements.Q: What industries can benefit from these validation testing services?
A: Industries like pharmaceuticals, healthcare, biotechnology, and manufacturing, where compliance and data integrity are crucial, gain significant advantages from our meritorious validation testing solutions.Q: What is the typical process for engaging your CSV Testing Services?
A: The process starts with an initial consultation and review of your system requirements. We then plan the validation activities, execute testing with customized or industry-standard tools, and provide complete documentation for regulatory submission.Q: When can clients expect delivery of validation reports and protocols?
A: Project timelines are tailored to client needs, and we ensure efficient handover of validation protocols, test reports, and SOPs upon project completion, consistent with agreed delivery schedules.Q: Where are your services available and how are they provided?
A: Our CSV Testing Services are available pan India and can be delivered both online and offline, offering flexibility to suit diverse client requirements.Q: What support is offered throughout the validation lifecycle?
A: Clients receive end-to-end support, from project planning and risk assessment to post-validation maintenance and ongoing compliance assistance, all with strict confidentiality.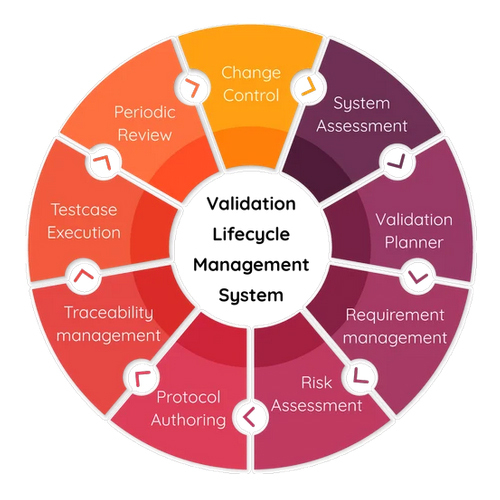
Tell us about your requirement

Price:
Quantity
Select Unit
- 50
- 100
- 200
- 250
- 500
- 1000+
Additional detail
Mobile number
Email







 Send Inquiry
Send Inquiry Send SMS
Send SMS Call Me Free
Call Me Free